Dual-Site Models
In this case study, we study ethylene epoxidation via a dual site microkinetic model. The microkinetc model entails only 4 elementary reaction steps:
- ethylene adsorbs on site family
S1 - oxygen adsorbs dissociatively on site family
S2 - one
S1adsorbate reacts with oneS2adsorbate - the product remains on
S1and desorbs fromS1
Mechanism
The four elementary steps are:
The first, second, and fourth steps are single-site gas/surface exchange
steps. The third step is a pair reaction because it requires one occupied S1
site and one occupied S2 site.
Example Input
let s1 = {site="S1", site_density=1.0}
let s2 = {site="S2", site_density=1.0}
let topo = PairStatistics(pair_densities={S1-S1=1.0, S1-S2=0.5, S2-S2=1.0})
components {
E {phase=gas, init=1.0, role=reactant},
O2 {phase=gas, init=1.0, role=reactant},
EO {phase=gas, init=0.0, role=product},
*S1 {phase=surface, init=1.0, tags=[emptysite], *s1},
E*S1 {phase=surface, init=0.0, *s1},
EO*S1 {phase=surface, init=0.0, *s1},
*S2 {phase=surface, init=1.0, tags=[emptysite], *s2},
O*S2 {phase=surface, init=0.0, *s2},
}
reactions {
{E}+{*S1}=>{E*S1}
@ HertzKnudsenDefault(Asite=4.43e-28, m=28, theta=2.8, sigma=1, S=1, Edes=92e3),
{O2}+2{*S2}=>2{O*S2}
@ HertzKnudsenDefault(Asite=2.36e-28, m=32, theta=2.08, sigma=2, S=1, Edes=95e3),
{E*S1}+{O*S2}=>{EO*S1}+{*S2}
@ ArrheniusDefault(Vf=1e13, Vb=1e13, Eaf=124441, Eab=240000)
@ PairMeanField(topology=topo),
{EO}+{*S1}=>{EO*S1}
@ HertzKnudsenDefault(Asite=5.55e-28, m=44, theta=2.56, sigma=1, S=1, Edes=95e3)
}
Only the pair reaction uses the topology attachment. The single-site steps use their ordinary kinetic laws unchanged.
General Rate Equations
This case study allows for an analytical solution, which is derived below
- \(\theta_E\) be the coverage of
E*S1 - \(\theta_{EO}\) be the coverage of
EO*S1 - \(\theta_O\) be the coverage of
O*S2
with the site balances
and site densities
for S1 and S2.
For the pair step we also need the mean-field abundance of S1-S2 neighbor
pairs:
The topology-aware elementary rates are then
The steady-state coverage equations become
These equations already show why the site ratio can now change the final EO rate: \(P_{12}\) appears directly inside the steady-state chemistry instead of only in the transient scaling.
Reduced Analytical Form
For the present benchmark \(p_E = 1\), \(p_{O_2} = 1\), and \(p_{EO} = 0\), so step 4 contributes only desorption. Writing
and defining
the steady-state equations are
When the backward cross term is negligible, this simplifies to
and
Introduce
and
Then the remaining oxygen balance reduces to the cubic
This cubic can have up to three mathematical roots, but only one is physically relevant: the root for which
and, after back-substitution,
So the analytical construction is:
- solve the cubic for \(\theta_O\)
- keep the single physical root
- recover \(\theta_E\) and \(\theta_{EO}\) from the relations above
The reproduction script solves the full steady-state system numerically rather than this reduced cubic, but it is solving the same analytical branch and comparing that branch against the numerical MKMCXX result for every temperature and pair density.
Linking Site Density to Pair Density
Site density and pair density are connected, but not identical.
- \(\Gamma_1\) and \(\Gamma_2\) tell us how many
S1andS2sites exist. - \(P_{12}\) tells us how many
S1-S2pairs exist.
Two surfaces can have the same S1:S2 ratio and still have different
\(P_{12}\) if one is highly intermixed and the other is segregated.
For this case study we use the simplest random-mixing closure:
With \(\Gamma_1 = 1\), this gives
So in this benchmark the extreme 1:10 site-ratio case actually has fewer
mixed S1-S2 pairs than the 1:1 case, which lowers the EO formation rate.
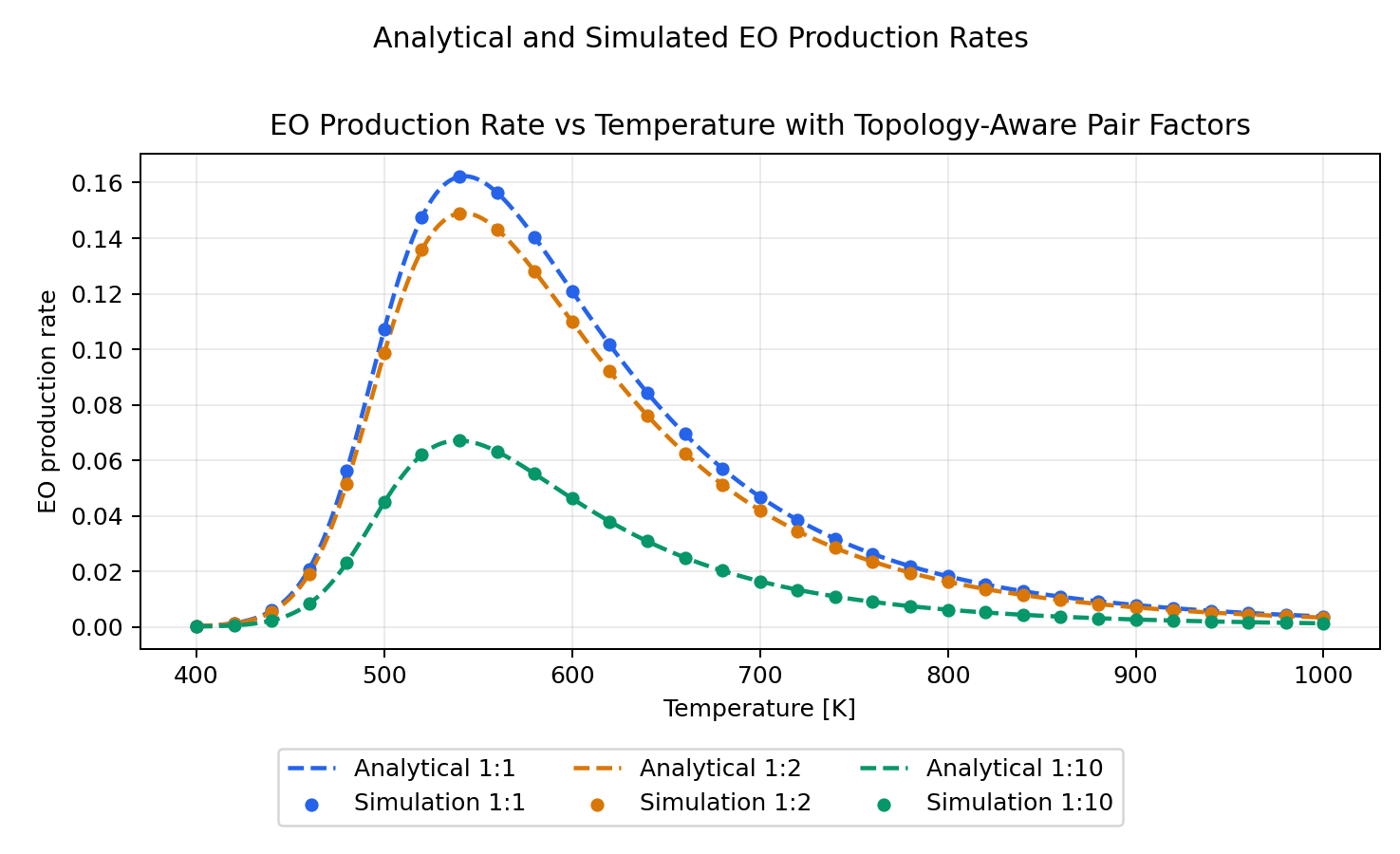
Numerical Check
We explore here three site-ratio cases:
S1:S2 = 1:1S1:S2 = 1:2S1:S2 = 1:10
For each case it
- computes \(P_{12}\) from the random-mixing closure
- generates a materialized
.mkxinput withPairStatistics(...) - solves the analytical steady-state equations across
400-1000 K - runs the numerical MKMCXX simulation across the same temperatures
- compares the EO production rate from both
At 500 K, the reproduced EO production rates are:
S1:S2 |
\(P_{12}\) | Analytical EO rate | Simulated EO rate |
|---|---|---|---|
1:1 |
0.500000 |
0.107178 |
0.107178 |
1:2 |
0.444444 |
0.0985688 |
0.0985688 |
1:10 |
0.165289 |
0.0448396 |
0.0448396 |
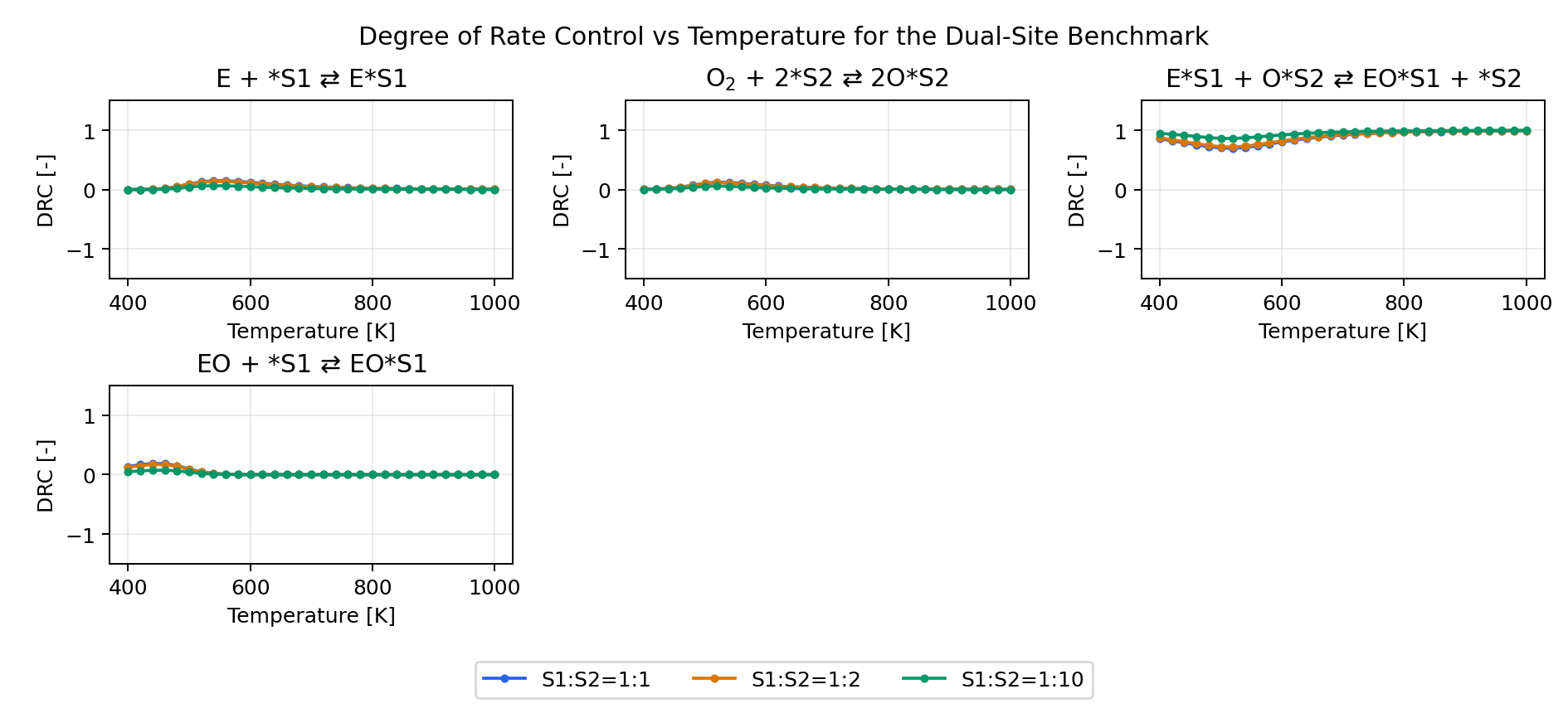
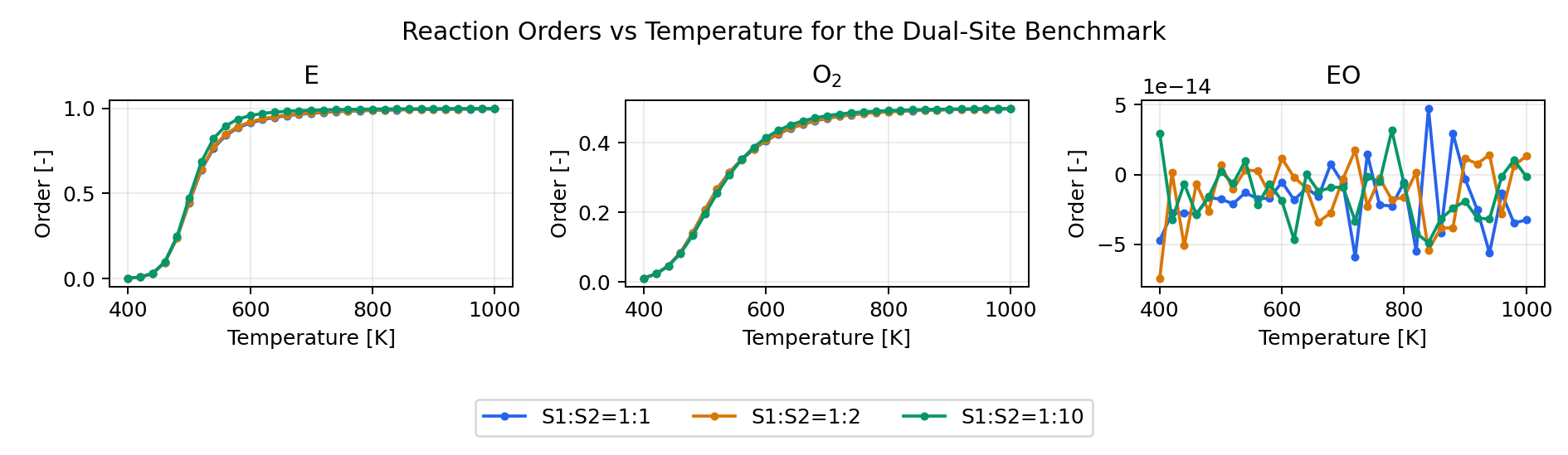
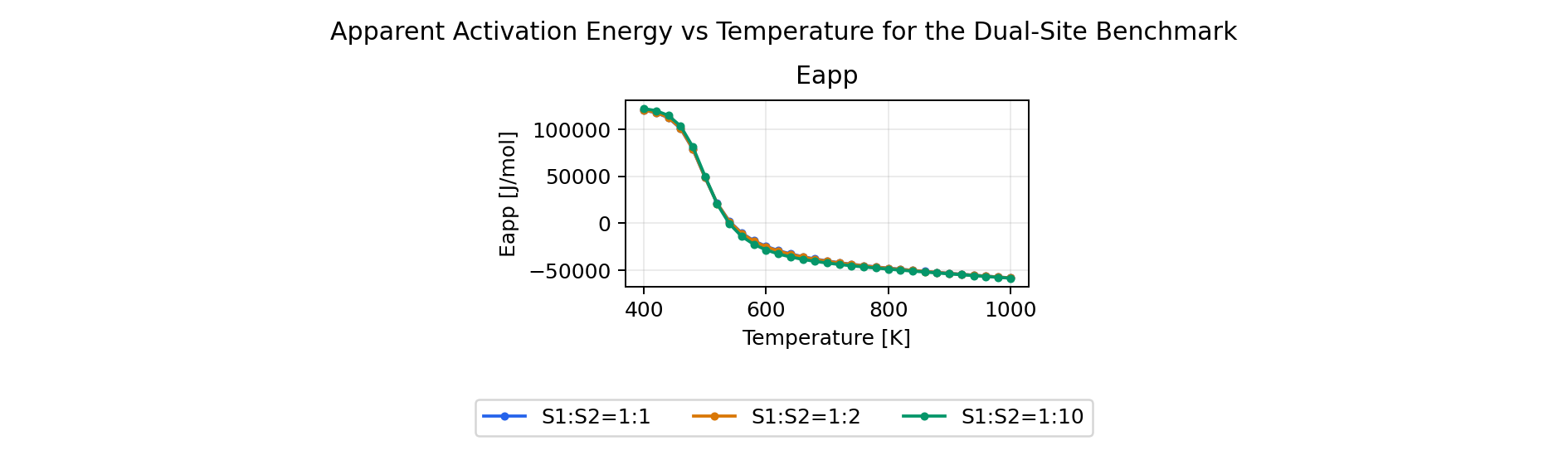
Derived Analyses
The same benchmark input also enables the derived analyses:
- reaction orders
- apparent activation energy
- degree of rate control
- thermodynamic degree of rate control
To make those analyses meaningful, the example tags EO as the key product,
marks the surface species for tdrc, and applies drc tags to the elementary
steps. The scenario script then produces the analysis outputs for the same
1:1, 1:2, and 1:10 site-ratio cases.
Reaction Orders
Apparent Activation Energy
Degree of Rate Control